Technologies
Learn more about the technology that is behind out MenlaScan Systems products.
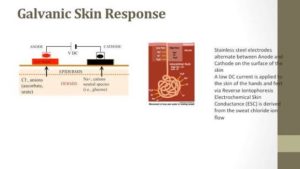
Galvanic Skin Response (GSR) is defined as the change in the electrical properties of the skin. The signal test can be used for capturing the autonomic nerve responses as a parameter of the sweat gland function. The measurement is relatively simple and repeatable, making the GSR assessment a useful tool for examination of the autonomous nervous system function, and especially the peripheral sympathetic system.
Several terms are used for this phenomenon, such as EDA (electrodermal activity), EDR (electrodermal response), EDL (electrodermal level), SCA (skin conductance activity), SCR (skin conductance response), including more.
Galvanic Skin Response can be described in terms of conductance, resistance, and electrophysiological potential. The electro-physiological signal is generated by the sweat glands, where sweat is likely the origin of the variation in resistance and conductivity, although the vaso- dilatation and constriction may also play an important role.
In most cases, the GSR is measured on areas of skin with high sweat gland concentration. As a reference, you can use a part of the skin with less or no sweat glands, or measure in the same area as the active electrode.
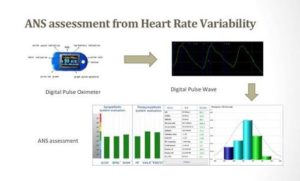
Heart Rate Variability’ is the conventionally accepted term to describe oscillations in the interval between both consecutive heart beats and consecutive instantaneous heart rates. In order to describe oscillation in consecutive cardiac cycles, other terms have been used in the literature, for example cycle length variability, heart period variability, RR variability and RR interval tachogram, and they more appropriately emphasize the fact that it is the interval between consecutive beats that is being analyzed rather than the heart rate per sec.
During the 1970s, Ewing et al. devised a number of simple bedside tests of short-term RR differences to detect autonomic neuropathy in diabetic patients. The association of higher risk of post-infarction mortality with reduced HRV was first shown by Wolf et al. In 1977. In 1981, Akselrod et al. introduced power spectral analysis of heart rate fluctuations to quantitatively evaluate beat-to-beat cardiovascular control.
These frequency–domain analyses contributed to the understanding of the autonomic background of RR interval fluctuations in the heart rate record. The clinical importance of HRV became apparent in the late 1980s when it was confirmed that HRV was a strong and independent predictor of mortality following an acute myocardial infarction. With the availability of new, digital, high frequency, 24-h multi-channel electrocardiographic recorders, HRV has the potential to provide additional valuable insight into physiological and pathological conditions and to enhance risk stratification.
Variations in heart rate may be evaluated by a number of methods. Perhaps the simplest to perform are the time domain measures. With these methods either the heart rate at any point in time or the intervals between successive normal complexes are determined. In a continuous electrocardiographic (ECG) record, each QRS complex is detected, and the so-called normal-to-normal (NN) intervals (that is all intervals between adjacent QRS complexes resulting from sinus node depolarizations), or the instantaneous heart rate is determined. Simple time–domain variables that can be calculated include the mean NN interval, the mean heart rate, the difference between the longest and shortest NN interval, the difference between night and day heart rate, etc. Other time–domain measurements that can be used are variations in instantaneous heart rate secondary to respiration, tilt, Valsalva manoeuvre, or secondary to phenylephrine infusion. These differences can be described as either differences in heart rate or cycle length.
The autonomic nervous system response to athletic training and rehabilitative exercise programs after various disease states is thought to be a conditioning phenomenon. HRV data should be useful in understanding the chronological aspects of training and the time to optimal conditioning as it relates to the autonomic influences on the heart.
Digital Pulse Wave Analysis
Analysis of the contour of the peripheral pulse to assess arterial properties was first described in the nineteenth century. With the recognition of the importance of arterial stiffness there has been a resurgence of interest in pulse wave analysis, particularly the analysis of the radial pressure pulse acquired using a tonometer. An alternative technique utilizes a volume pulse. This may conveniently be acquired optically from a finger (digital volume pulse). Although less widely used, this technique deserves further consideration because of its simplicity and ease of use.
As with the pressure pulse, the contour of the digital volume pulse is sensitive to changes in arterial tone induced by vasoactive drugs and is influenced by ageing and large artery stiffness. Measurements taken directly from the digital volume pulse or from its second derivative can be used to assess these properties. The arterial pulse waveform is a contour wave generated by the heart when it contracts, and it travels along the arterial walls of the arterial tree. Generally, there are 2 main components of this wave: forward moving wave and a reflected wave.
The forward wave is generated when the heart (ventricles) contracts during systole. This wave travels down the large aorta from the heart and gets reflected at the bifurcation or the across-road of the aorta into 2 iliac vessels. In a normal healthy person, the reflected wave usually returns in the diastolic phase, after the closure of the aorta valves. The returned wave give a notch and it also helps in the perfusion of the heart through the coronary vessels as it pushes the blood through the coronaries.
Therefore the velocity at which the reflected returns becomes very important: the stiffer the arteries are, the faster it returns. This may then enter into the systolic phase and augment final blood pressure reading.
link to Scientific Background which will have the following research papers you can find here
Bio Impedance & Body Composition
Electrical impedance is the opposition to the flow of an electrical current, being the ratio between an alternating sinusoidal voltage and an alternating sinusoidal flow. In consequence impedance is a passive magnitude that does not irradiate energy, therefore energy must be provided in two ways: by exciting the tissue with current or with voltage. In this case, the EBI measurement method used consists on injecting an electrical signal with a known current and measuring the reciprocal complementary voltage magnitude the value is calculated using the Ohm’s Law, in the frequency domain. Z = V / I
Where Z is the impedance, V the voltage and I the current. The diagram block of a typical impedance measurement system is the following: Typical diagram block of a impedance measurement system
Measurement types
Depending on the type of information that we are looking for analyse several types of measurements can be done. Non-phase BIA measurement: This method measures the total resistance of the body (Z). This does not give determination of the phase angle and, as such, a subdivision of impedance into water and cellular resistance, so that no judgement can be made about the body cell mass or the extra-cellular mass with non-phase measurements Phase BIA measurement: The phase sensitive technique of measuring allows the impedance Z to be differentiated into its two components resistance (R), that shows the water resistance, and reactance (Xc), that show the cell resistance. This let us differentiate between the body cell mass and extra-cellular mass. Phase BIA multi-frequency measurement: BIA is frequency dependent, therefore for a more intensive study we can study the resistance and reactance in a different single low frequencies measurements (1-5 KHz). With this type of measurement it is possible to achieve a subdivision of the total body water into intra-cellular and extra-cellular body water.
Measurement Techniques
There are several approaches to measure Bioimpedance that should be chosen depending on the desired characteristics of the system built. Null Techniques: Detection with this technique is very simple and it is based in a simple ampere meter. The most common method used is a Wheatstone bridge. This is a high accurate method with the inconvenient of needing a large number of electronic components and not being time efficient in some applications because of its iterating process. Deflection Techniques: The impedance estimation is done by measuring the voltage drop or the current through the load as a response to an alternative known current. This method is based on simple electronics using complex operations. Due to Measurements of Bioimpedance & Instrumentation this an application specific integrated circuit or microcontroller is needed to carry these operations. Its main characteristic is the time efficient, being able to do short time accuracy measurements. For the impedance estimation we are going to focus only in the deflection techniques that are those that are commonly used in bioimpedance measurements. In impedance estimation is it possible to do single frequency and multy-frequency analysis using different techniques, basing the study in the known excitation provided and the measured obtained. So for single frequency or sweep frequency measurements usually Sine Correlation is used and for multi-frequency measurements it is common used the Fourier Transform.
Renal function monitoring
Electrical Bioimpedance measurements have been used for many years to study the electrical properties of biologic tissue and to measure physiological events, being applied in several clinical areas, including body composition. The assessment of oedema formation from EBI measurements is based on the dependency of the electrical properties of tissue on its structure and intrinsic constituents, i.e. alterations in the tissue structure produce a modification of its electrical properties.
When the renal function is impaired in the kidneys, an immediate physiological consequence is an excessive accumulation of liquid in the rest of the body, and consequently oedema is formed. Oedema is considered by the physician a common indicator of renal failure, and therefore visual inspection by the physician is a regular practice to detect such kidney function impairment. Such an inspection is usually performed targeting the limbs, legs and arms, targeting the inspection for peripheral oedema.
There are several methods of assessing extracellular swelling, and the EBI measurement approach is one of the more comfortable ones, due to the fact that the electrical properties of tissue can be measured non-invasively and without tightening the skin. Some studies about the excess of fluid in chronic HD patients, using Bioimpedance spectroscopy measurements, where performed. The experimental results suggested that HD patients keep their excess fluid volume primarily in the extracellular compartment (interstitial fluid). Bioimpedance spectroscopy together with a stable measurement of lean tissue can determine the degree of relative excess hydration. Due to the capacitance effects of the cell membrane, the tissue impedance depends on the measurement frequency. As a consequence, the accuracy of measurements by means of multiple frequency Bioimpedance spectroscopy analysis is superior to the accuracy of measurements based on a single frequency for the prediction of extracellular water. Some authors suggest that the best frequency range to assess extra-cellular fluid is up to 10 kHz and the range between 50-100 kHz is a suitable measurement range for a successful assessment of extra- and intra-cellular fluid.
Concluding, the monitoring principle of the EBI measurement system implemented in this thesis work lays as follows: during renal failure the amount of interstitial fluid in the limbs increases, causing extracellular oedema. The consequent interstitial swelling modifies the electrical properties of the tissue, and by means of the combination of non- invasive EBI measurements with skin-surface electrodes and EBI spectroscopy analysis, the ongoing swelling can be detected. Therefore changes in the EBI of the limbs may be used as an efficient indicator for early detection of renal failure.
Lung edema
Symptoms of lung edema usually appear at a very late stage when the amount of fluid in the lungs has already sextupled.
At this stage, the patient typically has dyspnea (short breath) and hypoxia (low partial pressure of oxygen in the blood) because of impaired gas exchange due to the increased interstitial fluid volume. As a consequence, the patient is usually committed to an intensive care unit for medical treatment. Preclinical detection and continuous monitoring of the lung fluid volume during medical treatment would enhance medical care while reducing costs.
Unfortunately, conventional detection methods like radiographic imaging, monitoring pulmonary capillary wedge pressure or double indicator thermodilution are impractical for continuous monitoring. A promising alternative is the Bioimpedance Spectroscopy (BIS). Detection of lung edema using BIS is based on the fact that the amount of fluid in the lungs has significant impact on their electrical impedance. Normally, the lungs have about 5% of fluid and 95% of air, resulting in an electrical impedance in the range of about 10 to 20 Ohm. If the amount of fluid in the lungs increases, electrical impedance decreases because of the much lower electrical impedance of fluid (serum, for example, has a resistance of about 0.6 Ohm).
Compared to other measurement methods, BIS has two significant advantages: the measurement is non-invasive and it can easily be done at the patient’s home. In previous work, BIS was shown to be practical for detection of lung edema and, in a single-frequency version, it is used in some ICDs (implantable cardioverter-defibrillator).
Bio Impedence and Body Composition
Bioelectrical impedance analysis (BIA) is a widely used technique for estimating body composition and it is particularly useful in large, population-based studies because it is quick, portable, inexpensive and non-invasive. Using the resistance and reactance measurements of the human body in tetrapolar mode and in multi-frequency from 1 to 500 KHz, the appropriate algorithms issue from the peer reviews will allow the estimation of the body composition.
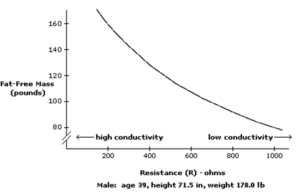
In the human body, low resistance is associated with large amounts of fat-free mass and low fat mass. High resistance is associated with smaller amounts of fat-free mass and high fat mass.
In the human body, high reactance is associated with large amounts of body cell mass (intracellular mass) and low ECW (Extra Cellular Water). Low reactance is associated with smaller amounts of body cell mass and high ECW.